Ligustrum spp. TS safranin-o stain
Posted: Thu Jul 29, 2021 10:42 pm
Hi all,
Recently I began botanical sectioning, and I give my thanks to forum members mrsonchus and MicroBob for their excellent and very helpful posts on this subject matter. After experimenting with various plants nearby, I found that the privet bush (Ligustrum spp.) happens to be a particularly excellent plant for hand sectioning. Thus begins my little experiment in hand sectioning and staining transverse sections of Ligustrum vulgare (?)
1 - background
Ligustrum is a tall and resilient hedge often used in gardens, but is sometimes a small tree depending on the species. They flower and produce berries (unfortunately I missed the flowering period this year and did not get a chance to section the flowers ... next summer!)
... next summer!)

An image of Ligustrum vulgare which I sectioned.

The branches of Ligustrum have multiple stages of growth. Prior to becoming a tough and woody branch once it is fully grown (see image above - branch 3), the young, sprouting branches have a green color and are strong, but much softer than the fully-grown branch (see image above - branches 1 and 2). I have found that the privet branch at this sprouting stage is optimal for hand sectioning; it is soft enough that a razor blade cuts through without much effort, but thick and strong enough that thin sections and the pressure of cutting still allow the section to retain its shape and not get crushed.
2 - procedures
A single edge razor blade was utilized for sectioning (a method that requires much practice and patience! ) and as I was making a temporary mount because my main objective was to test staining, I opted for a pva glue and glycerin mounting medium. It is a 1:1 mixture of a pva-based glue (Elmer's clear glue, for example) and glycerin, along with some water. This recipe is derived from Richard H. Zander's 2014 paper linked here: http://www.mobot.org/plantscience/ResBo ... ia2014.pdf. It is a useful mountant: easy to make and use, and preserves the slides for many months.
) and as I was making a temporary mount because my main objective was to test staining, I opted for a pva glue and glycerin mounting medium. It is a 1:1 mixture of a pva-based glue (Elmer's clear glue, for example) and glycerin, along with some water. This recipe is derived from Richard H. Zander's 2014 paper linked here: http://www.mobot.org/plantscience/ResBo ... ia2014.pdf. It is a useful mountant: easy to make and use, and preserves the slides for many months.
I tested 2 different staining protocols to see the difference in results. Protocol 1, derived from mrsonchus' post here: https://www.microbehunter.com/microscop ... 10&t=7889&: 1% safranin-o aqueous for 5 seconds, transfer sections to water for a bit to remove excess stain, then transfer sections to 70% ethyl alcohol for 30 minutes. Mrsonchus did 50% alcohol to 95% alcohol - I only had 70% alcohol on hand and it seemed to still work. Protocol 2, derived from mrsonchus' post here: https://www.microbehunter.com/microscop ... 1&p=44944&: sections in a weak (diluted) safranin-o stain for 1 hour.
3 - results
These were my most successful sections and staining so far - for a beginner like me this makes me very happy!
Protocol 1:

40x - quite possibly the best section in this batch - I think it took to the stain well, the xylem are particularly resolvable and so are the medullary parenchyma in the middle.

xylem, 100x

medullary parenchyma, 100x

xylem, 400x

40x, protocol 1 but a different section - it took to the stain differently despite enduring the same conditions - not sure why

100x, medullary parenchyma


collenchyma, 100x and 400x
Protocol 2:

40x, a much deeper red than resulted in protocol 1, still gorgeous though. But the difference in protocols definitely shows
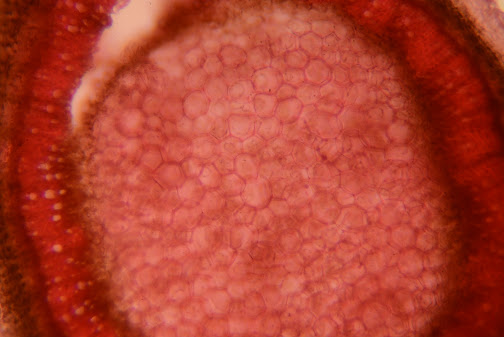
100x, medullary parenchyma and xylem
4 - conclusion
That concludes my little experiment. I had a lot of fun and consider this to be one of my first bigger successes with botanical sectioning. Happy to see the safranin working well and my hand sectioning skills improving. I look forward to investing in other stains - possibly fast green or alcian blue to be able to follow more complex staining procedures. I did this all with very simple materials - hopefully this shows that botanical sectioning is not as daunting as it seems and even beginners can achieve some decent sections - go out and try it if you haven't, it's tons of fun! I won't be forgetting about Ligustrum, but I look forward to my next adventure: pollen!
Recently I began botanical sectioning, and I give my thanks to forum members mrsonchus and MicroBob for their excellent and very helpful posts on this subject matter. After experimenting with various plants nearby, I found that the privet bush (Ligustrum spp.) happens to be a particularly excellent plant for hand sectioning. Thus begins my little experiment in hand sectioning and staining transverse sections of Ligustrum vulgare (?)
1 - background
Ligustrum is a tall and resilient hedge often used in gardens, but is sometimes a small tree depending on the species. They flower and produce berries (unfortunately I missed the flowering period this year and did not get a chance to section the flowers

An image of Ligustrum vulgare which I sectioned.

The branches of Ligustrum have multiple stages of growth. Prior to becoming a tough and woody branch once it is fully grown (see image above - branch 3), the young, sprouting branches have a green color and are strong, but much softer than the fully-grown branch (see image above - branches 1 and 2). I have found that the privet branch at this sprouting stage is optimal for hand sectioning; it is soft enough that a razor blade cuts through without much effort, but thick and strong enough that thin sections and the pressure of cutting still allow the section to retain its shape and not get crushed.
2 - procedures
A single edge razor blade was utilized for sectioning (a method that requires much practice and patience!
I tested 2 different staining protocols to see the difference in results. Protocol 1, derived from mrsonchus' post here: https://www.microbehunter.com/microscop ... 10&t=7889&: 1% safranin-o aqueous for 5 seconds, transfer sections to water for a bit to remove excess stain, then transfer sections to 70% ethyl alcohol for 30 minutes. Mrsonchus did 50% alcohol to 95% alcohol - I only had 70% alcohol on hand and it seemed to still work. Protocol 2, derived from mrsonchus' post here: https://www.microbehunter.com/microscop ... 1&p=44944&: sections in a weak (diluted) safranin-o stain for 1 hour.
3 - results
These were my most successful sections and staining so far - for a beginner like me this makes me very happy!
Protocol 1:

40x - quite possibly the best section in this batch - I think it took to the stain well, the xylem are particularly resolvable and so are the medullary parenchyma in the middle.

xylem, 100x

medullary parenchyma, 100x

xylem, 400x

40x, protocol 1 but a different section - it took to the stain differently despite enduring the same conditions - not sure why

100x, medullary parenchyma


collenchyma, 100x and 400x
Protocol 2:

40x, a much deeper red than resulted in protocol 1, still gorgeous though. But the difference in protocols definitely shows

100x, medullary parenchyma and xylem
4 - conclusion
That concludes my little experiment. I had a lot of fun and consider this to be one of my first bigger successes with botanical sectioning. Happy to see the safranin working well and my hand sectioning skills improving. I look forward to investing in other stains - possibly fast green or alcian blue to be able to follow more complex staining procedures. I did this all with very simple materials - hopefully this shows that botanical sectioning is not as daunting as it seems and even beginners can achieve some decent sections - go out and try it if you haven't, it's tons of fun! I won't be forgetting about Ligustrum, but I look forward to my next adventure: pollen!